Clinical Application
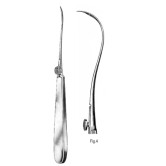
The Reverdin needle with a Fig.4 configuration is intended to facilitate suture passage in confined operative fields where traditional needle threading is difficult. Its openable eye allows the operator to capture and release suture without threading the suture end through a closed eye, reducing manipulation and tissue trauma. It is commonly used in delicate soft-tissue procedures such as ophthalmic, plastic/reconstructive, ENT, and hand surgery where controlled placement and minimal handling are required.
Design & Configuration
The Fig.4 curvature provides a controlled arc suited for accessing shallow-to-moderate depth pockets while maintaining predictable needle trajectory. The Reverdin (openable) eye mechanism permits single-handed loading and release of suture within narrow exposure sites, eliminating the need to pass bulky needle holders or to externalize long suture tails. At 19 cm overall length, the shaft offers reach and fingertip control for precise placement in confined anatomical corridors.
Instrument Specifications
- Instrument type: Reverdin needle
- Pattern / model: Fig.4
- Configuration: Openable (Reverdin) eye, curved configuration
- Length: 19 cm
- Tip / jaw pattern: Precision needle point with Reverdin (split/open) eye for suture capture
- Typical use: Passage and placement of suture in delicate or confined soft-tissue procedures
- Material: Surgical-grade stainless steel
- Reusable
Material & Construction
Manufactured from surgical-grade stainless steel with a precision-formed needle point and a machined open-eye capture mechanism for reliable suture engagement. The instrument is designed for repeated use and is compatible with standard cleaning and sterilization processes used for reusable stainless-steel surgical instruments.
Quality & Compliance
Manufactured by FDA-registered OEM facilities and produced under strict quality control standards.
Brand Comparison
Comparable in form and function to equivalent instruments offered by major US medical suppliers.
Availability & Returns
- In-stock lead time: ships within ~1 week
- Out-of-stock lead time: 2–4 weeks
- Returns: 30-day return policy




































