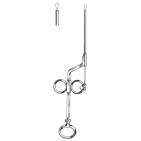
Clinical Application
Designed for controlled excision of intranasal soft tissue, the Glegg nasal snare is used primarily for polypectomy and removal of pedunculated nasal lesions under endoscopic or direct visualization. The cutting wire loop permits atraumatic capture of a polyp stalk and progressive tightening to transect tissue while minimizing collateral mucosal trauma. It is appropriate for use in clinic-based procedures and operating room settings where mechanical resection of benign intranasal masses is indicated.
Design & Configuration
The instrument’s cannulated shaft guides a single wire loop that is deployed at the distal tip and controlled by an ergonomic sliding-handle mechanism. This geometry provides tactile feedback and precise control of loop closure, allowing the operator to place the loop around a lesion’s stalk and incrementally tighten to achieve clean transection. The narrow shaft profile facilitates access to the narrow nasal cavity and paranasal recesses while the protected cannula helps shield adjacent mucosa during placement.
Instrument Specifications
- Instrument type: Nasal snare (cutting)
- Pattern / model: Glegg
- Configuration: Cannulated shaft with single-loop wire and sliding-handle control
- Tip / jaw pattern: Single wire-loop cutting snare
- Typical surgical use: Endoscopic intranasal polypectomy and excision of soft nasal masses
- Material / construction: Surgical-grade stainless steel shaft with stainless wire loop
- Reusable: Designed for repeated sterilization and reuse
Material & Construction
Constructed with surgical-grade stainless steel for durability and corrosion resistance, the device uses a resilient stainless wire loop sized for controlled cutting. The design supports repeated high-level cleaning and sterilization cycles common in ENT instrument workflows. No disposable electronics or fiber optic components are required for operation.
Quality & Compliance
Manufactured by FDA-registered OEM facilities and produced under strict quality control standards.
Brand Comparison
Comparable in form and function to equivalent instruments offered by major US medical suppliers.
Availability & Returns
- In-stock lead time: ships within ~1 week
- Out-of-stock lead time: 2–4 weeks
- Returns: 30-day return policy




































