Clinical Application
Designed for use in obstetric and gynecologic procedures where controlled grasping and removal of soft intrauterine tissue is required. The instrument is intended for manual removal of the placenta and retained products of conception, retrieval of ovum or fetal tissue fragments, and for procedures that require atraumatic secure handling of membranes and friable tissue within the uterine cavity. Its length and jaw geometry help the operator reach and extract tissue with direct control while minimizing slippage.
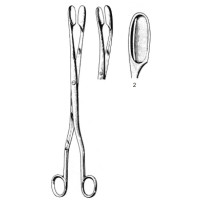
Design & Configuration
The straight shaft provides a direct line of access to the uterine cavity, facilitating reach and maneuverability in the third stage of labor or during operative evacuation. The Winter-style, Figure 2 jaw profile features broad, cupped surfaces that distribute pressure across a larger contact area to improve grip on soft, slippery tissue while reducing focal compression. The 28 cm overall length balances intracavitary reach with tactile feedback in the surgeon's hand for controlled traction and extraction.
Instrument Specifications
- Instrument type: Forceps
- Pattern / model: Winter (Figure 2)
- Configuration: Straight
- Length: 28 cm
- Tip / jaw pattern: Broad, cupped jaws for secure handling of placental tissue and membranes
- Typical surgical use: Removal of placenta, retained products of conception, ovum retrieval
- Material / construction: Surgical stainless steel; reusable
Material & Construction
Manufactured from surgical-grade stainless steel with precision-formed jaws to maintain consistent alignment and grasp through repeated use. The construction is intended for repeated sterile processing and is compatible with standard autoclave sterilization protocols for reusable instruments.
Quality & Compliance
Manufactured by FDA-registered OEM facilities and produced under strict quality control standards.
Brand Comparison
Comparable in form and function to equivalent instruments offered by major US medical suppliers.
Availability & Returns
- In-stock lead time: ships within ~1 week
- Out-of-stock lead time: 2–4 weeks
- Returns: 30-day return policy




































