Clinical Application
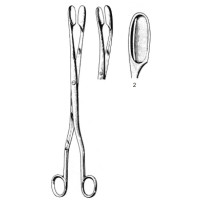
Long, curved placenta and ovum forceps designed for obstetric and gynecologic procedures where secure retrieval of soft tissue is required. Common clinical uses include controlled manual removal of placenta, extraction of retained products of conception following delivery or miscarriage, and targeted retrieval of ovum or tissue from the uterine cavity and vaginal canal. The length and curvature facilitate reach into the uterine cavity while allowing the operator to maintain control at the external opening of the vagina.
Design & Configuration
The instrument’s 28 cm working length provides extended reach for intrauterine access without excessive hand intrusion. The curved (cvd) shaft with a Fig.2 tip creates a gentle anatomic profile that follows the uterine axis, improving alignment with tissue planes and helping reduce blind grasping. Cupped/fenestrated jaws deliver a secure hold on friable placental or ovum tissue while distributing compressive force to minimize fragmentation during extraction.
Instrument Specifications
- Instrument type: Forceps
- Pattern / model: Winter placenta and ovum forceps, Fig.2
- Configuration: Curved (cvd)
- Length: 28 cm
- Tip / jaw pattern: Cupped / fenestrated jaws suitable for soft-tissue retrieval
- Typical surgical use: Retrieval of placenta, retained products of conception, and ovum
- Material / construction: Surgical stainless steel; reusable
Material & Construction
Constructed from surgical-grade stainless steel for durability and corrosion resistance in repeated clinical use. The design supports standard sterilization and reprocessing protocols; the instrument is reusable and intended for multiple cycles of cleaning and autoclave sterilization when processed according to facility guidelines.
Quality & Compliance
Manufactured by FDA-registered OEM facilities and produced under strict quality control standards.
Brand Comparison
Comparable in form and function to equivalent instruments offered by major US medical suppliers.
Availability & Returns
- In-stock lead time: ships within ~1 week
- Out-of-stock lead time: 2–4 weeks
- Returns: 30-day return policy




































